The XEMBIFY Patient Starter Kit includes:

Quick Reference Infusion Guide
Follow the steps, illustrated on this easy-to-read mat, to prepare and administer your infusions. Its water-resistant surface can be cleaned easily for repeated use.
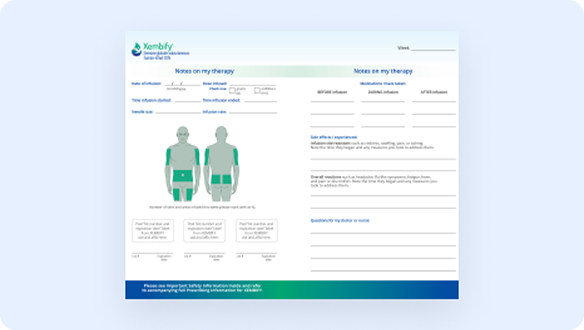
Infusion Log Book
Track your infusions and how they feel, and remember to share your Infusion Log Book with your healthcare provider.

Buzzy® Mini Personal
The Buzzy® Mini Personal device uses a patented combination of cold and vibration to reduce the pain of injections.



